 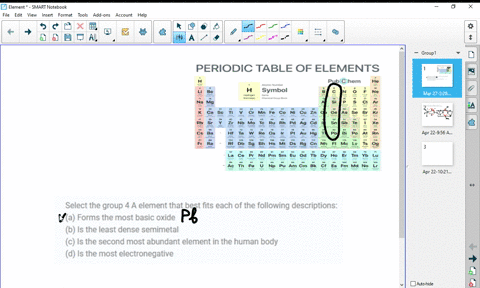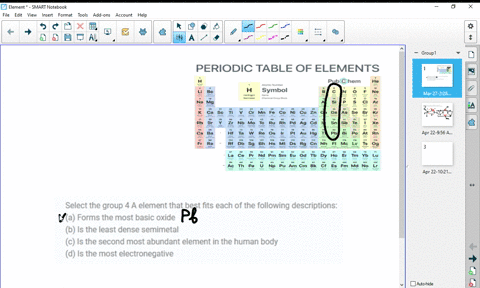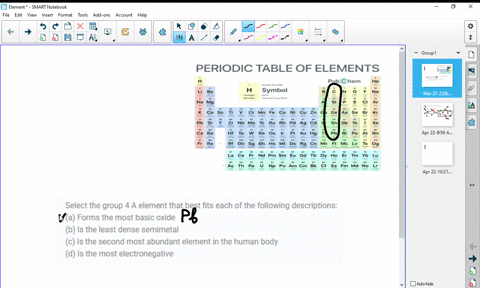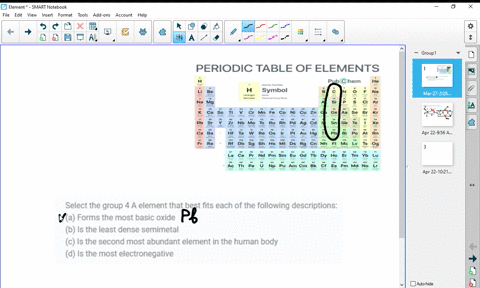
At higher temperatures, all three metals react with oxygen, nitrogen, carbon, boron, sulfur, and silicon. For the first three metals, it has been shown that they are resistant to concentrated alkalis, but halogens react with them to form tetrahalides. HĪs tetravalent transition metals, all three elements form various inorganic compounds, generally in the oxidation state of +4. The oxides TiO 2, ZrO 2 and HfO 2 are white solids with high melting points and unreactive against most acids. The reactivity is not always obvious due to the rapid formation of a stable oxide layer, which prevents further reactions. All the elements of the group are reactive metals with a high melting point (1668 ☌, 1855 ☌, 2233 ☌). The chemistry of rutherfordium is not very established and therefore the rest of the section deals only with titanium, zirconium, and hafnium. Most of the chemistry has been observed only for the first three members of the group. :max_bytes(150000):strip_icc()/the-periodic-table--digital-illustration--73016803-598b218ec41244001024af78.jpg)
Like other groups, the members of this family show patterns in its electron configuration, especially the outermost shells resulting in trends in chemical behavior: Z So far, no experiments in a supercollider have been conducted to synthesize the next member of the group, either unpentquadium (Upq) or unpenthexium (Uph), and it is unlikely that they will be synthesized in the near future. All isotopes of rutherfordium are radioactive. However, the fourth element rutherfordium (Rf), has been synthesized in the laboratory none of its isotopes have been found occurring in nature. The first three members of the group share similar properties all three are hard refractory metals under standard conditions. The three Group 4 elements that occur naturally are titanium (Ti), zirconium (Zr) and hafnium (Hf). The group itself has not acquired a trivial name it belongs to the broader grouping of the transition metals. This group lies in the d-block of the periodic table. In the modern IUPAC nomenclature, Group 4 of the periodic table contains titanium ( Ti), zirconium ( Zr), hafnium ( Hf) and rutherfordium ( Rf). група хемијских елемената sh:4.The Group 4 elements are a group of chemical elements in the periodic table. skupina de:Titangruppe eo:Elemento de grupo 4 ko:4족 원소 it:Elementi del gruppo 4 lt:4-tos grupės elementai lmo:Grupp 4 de la taula periòdica mk:Елементи од група 4 nl:Titaangroep nn:Gruppe 4 sk:4. grupa hemijskih elemenata ca:Grup 4 de la taula periòdica cs:4. Solid borders are older than the Earth ( Primordial elements)ĭotted borders are made artificially ( Synthetic elements)Īr:عناصر المجموعة الرابعة ast:Elementos del grupu 4 bs:4. Rutherfordium's radioactivity would make it toxic to living cells.Įxplanation of right-hand periodic table slice: Titanium is one of the few first row d-block transition metals with no known biological role. They are hard refractory metals with low acqueous solubility and low availability to the biosphere. The group 4 elements are not known to be involved in the biological chemistry of any living systems. All isotopes of rutherfordium are radioactive, and have been synthesized in the laboratory, none of them have been found occurring in nature. Titanium's mechanical properties make it useful in alloys. These elements are hard refractory metals. In modern IUPAC nomenclature, Group 4 of the periodic table contains the following chemical elements: 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
